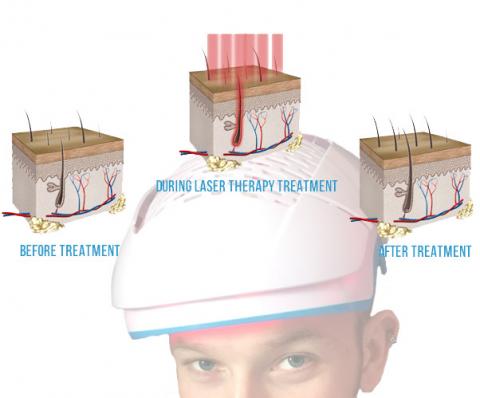
Low-Level Laser Therapy for Hair Loss Review, The Top 6 list
Having a bushy hipster beard has become a trend nowadays. For some people, growing beard requires a lot more than just letting it grow naturally. Minoxidil, being the no. 1 hair regrowth product, is used by many people to grow a beard. Many articles recommend Kirkland or Rogaine minoxidil for this purpose. Are they really the best minoxidil for bear growth? I would say 'no'. This blog will explain why.
Sign Up For Newsletter
We ship to UK, EU, North America, Canada, Japan, Australia and almost every country in the world.
Online store for all hair loss products. We ship to Austria, Italy, Belgium, Latvia, Bulgaria, japan, israel, Lithuania, Croatia, Luxembourg, Cyprus, Malta, portugal, Czechia, japan, new zealand, Netherlands, Denmark, without prescription, consultation, Poland, Estonia, Portugal, Finland, Romania, France, Slovakia, Germany, Slovenia, Greece, Spain, Hungary, Sweden, norway, Ireland
@ Copyright
by Minoxidilmax. All Rights Reserved.